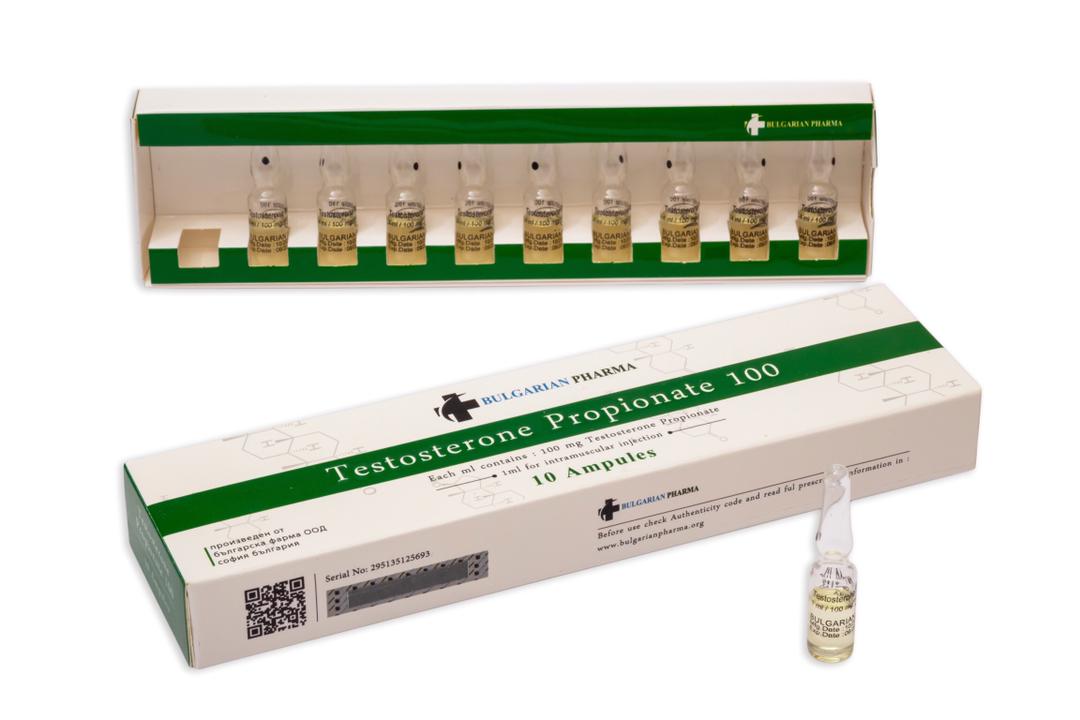
Testosterone Propionate 100
Active ingredients:Testosterone Propionate Pharmacological category:Androgenic hormone Composition:Every 1 ml contains 100 mg of Testosterone Propionate in Ethyl oleate Mechanism of medicine action: The effects of testosterone in humans and other vertebrates occur by way of two main mechanisms: by activation of the androgen receptor /directly or as DHT/, and by conversion to estradiol and activation of certain estrogen receptors. Free testosterone is transported into cytoplasm of target tissue cells, where it can bind to the androgen receptor, or can be reduced to 5-a di-hydrotestosterone by the cytoplasmic enzyme 5-alpha reductase. DHT binds to the same androgen receptor even more strongly than testosterone, so that its androgenic potency is about 5 times that of free testosterone. DHT-receptor complex under goes a structural change that allows it to move into the cell nucleus and bind directly to specific nucleotide sequences of the chromosomal DNA. The areas of binding are called hormone response elements, and influence transcriptional activity of certain genes, producing the androgen effects. Androgen receptors occur in many different vertebrate body system tissues, and both males and females respond similarly to similar levels. Greatly differing amounts of testosterone prenatally, at puberty, and throughout life account for a share of biological differences between males and females. Indications: Males: Testosterone Propionate is indicated for replacement therapy in conditions associated with a deficiency or absence of endrogenous testosterone: Primary hypogonadism testicular failure due to cryptorchidism, bilateral torsion orchitis, vanishing testis syndrome, or orchidectomy. Prologed androgen treatment will required to maintain sexual characteristics in these and other males who develop testosterone deficiency after puberty. Testosterone Propionate may be used to stimulate puberty in carefully selected males with clearly delayed puberty that is not secondary to a pathologic disorder; puberty is expected to occur spontaneously at a relatively late date. Brief treatment with conservative doses may occasionally be justified in these patients if they do not respond to psychologic support. The potential adverse effect on bone maturation should be discussed with the patient and parents prior to androgen administration. An x-ray of the hand and wrist to determine bone age should be obtained every 6 months to assess the effect of treatment on the epiphyseal centers. Females: Testosterone Propionate may be used secondarily in women with advancing inoperable metastatic mammary cancer who are 1 to 5 years postmenopausal. Primary goals of therapy in these women include ablation of the ovaries. Other methods of counteracting estrogen activity are adrenalectomy, hypophysectomy, and antiestrogen therapy. This treatment has also been used in premenopausal women with breast cancer who have benefited from ophorectomy and are considered to have a hormone responsive tumor. Judgement concerning androgen therapy should be made by an oncologist with expertisein this field. Testosterone Propionate has been used for the management of postpartum breast pain and engorgement. Contradictions: Testosterone Propionate is contradicted in men with carcinomas of the breast or with known or suspected carcinomas of the prostate and in women who are or may become pregnant. When administered to pregnant women, androgens cause virilization of the external genitalia of the female fetus. This virilization includes clitoromegaly, abnormal vaginal development, and fusion of genital folds to form a scrotal-like structure. The degree of masculinization is related to the amount of drug given and the age of the fetus and is most likely to occur in the female fetus when the drugs are given in the first trimester. If the patient becomes pregnant while taking these drugs, she should be apprised of the potential hazard of the fetus. Precautions: Testosterone Propionate, through its metabolic effects, stimulates the nervous, mental, and physical activities of a patient. Therefore, it should be used with caution in the presence of cardiovascular and renal diseases, especially in elderly male. Prolonged administration or excessive dosage may cause inhibition of testicular function. As a result, oligospermia may develop, and there may be a decrease in ejaculatory volume. Women should be observed for signs of virilization. Discontinuation of drug therapy at the time mild virilism becomes evident is necessary to prevent irreversible virilization. Such virilization usually follows administration of androgens at high doses. A decision may be made by the patient and the physician concerning the degree of virilization that will be tolerated during treatment for breast carcinoma. Anaphylactoid reactions, although rare, may occur, andtreatment should be readily available. Hypersensitivity reactions, including rash and dermatitis, have been reported. Drug interactions: Concurrent administration of oxyphenbutazone and androgens may result in elevated serum levels of oxyphenbutazone. In diabetic patients, the metabolic effects of androgens may decrease blood glucose and insulin requirements. Dosage: Testosterone Propionate is administered by intramuscular injection. It must not be given intravenously. Intramuscular injections should be given deep in the gluteal muscle. Males: the suggested dosage varies, depending on the age, sex, and diagnosis of the individual patient. Dosage is adjusted according to the patient's response and the appearance of adverse reactions, and maintenance doses should be the minimum that produce adequate effect. Replacement therapy in androgen-deficient males should be in the range of 25 to 50 mg every 2 to 3 times a week. Larger doses can be given by the doctor, depending of the case. For physique or performance enhancing purposes, the usual dose is in the range of 200-600 mg per week. Females: In inoperable carcinoma of the breast, temporary palliation may be obtained in some cases by therapy with androgens. A dosage of 50-100 mg of Testosterone Propionate administered intramuscularly 3 times weekly recommended. Storage: Store in a dark, dry place, at room temperature. Do not refrigerate. Keep out of reach of children. Manufactured by: Bulgarian Pharma Ltd.